- In vivo and in vitro Models /
- Research /
- Division: Laboratory Animal Science
Division – Laboratory Animal Science
Group leader: Prof. Dr. T. Kolbe
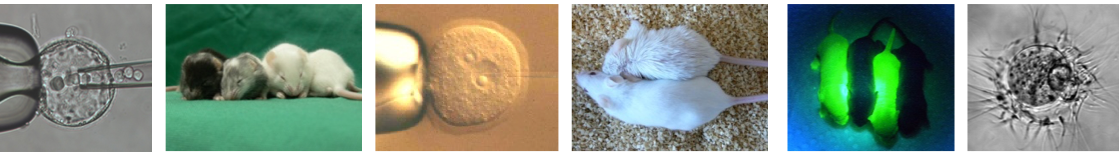
The research group Laboratory Animal Science focuses on mouse facility management and the generation of genetically modified mice. To generate knockout (KOs) or knockin (KIs) mouse lines we apply microinjection and blastocyst injection. In the laboratory classical cloning techniques are used to build gene constructs for DNA microinjection as well as targeting vectors for modifying murine embryonic stem cells via homologous recombination. The group is also using CRISPR/Cas9 technology for gene editing to generate much faster and more efficient knockout by DNA pronuclear microinjection. In addition, we also use CRISPR/Cas9 technology to establish faster and easier new embryonic stem cell lines for knockout/knockin mouse generation. The research group tries constantly to improve the methods used for gene editing and for developing genetically modified organisms.
A second important field of activity is the archiving of mouse lines and the provision of these mouse lines to scientists worldwide. For this purpose we cryopreserve sperm of mouse lines in liquid nitrogen. In addition, we further archive different embryonic stages (zygotes – blastocysts) and revitalize the archived mouse lines by in vitro fertilization (IVF) and embryo transfer. We revitalize also cryopreserved sperm and embryos for external institutions. Rederivation of contaminated mouse lines is another important service for the scientific community.
Additionally, we offer a wide range of reproductive technologies for research projects: IVF for fast colony expansion or to provide large mouse cohorts for experiments. Ovary transfer to rescue aging mouse lines and following cryopreservation of female gametes in situ in ovaries. Aggregation of embryos, electrofusion of embryonic cells, or cytoplasm transfer to answer specific questions in developmental biology.
Besides generating genetically modified mouse models the research group aims to improve the housing conditions of laboratory animals together with the 3Rs division. Based on the refinement principle of the 3Rs we assess the benefits and applicability of different environmental enrichments. Additionally, we provide an important contribution to the reduction principle of the 3Rs in cryopreserving and revitalizing mouse lines.
Publications:
1.Hedegger K, Algül H, Lesina M, Blutke A, Schmid RM, Schneider MR, Dahlhoff M. Unraveling ERBB network dynamics upon betacellulin signaling in pancreatic ductal adenocarcinoma in mice. Mol Oncol. 2020 Aug;14(8):1653-1669. doi: 10.1002/1878-0261.12699
2.Kolbe T, Walter I, Rülicke T. Influence of graft size, histocompatibility,and cryopreservation on reproductive outcome following ovary transplantation in mice. J Assist Reprod Genet. 2019 Dec;36(12):2583-2591. doi: 10.1007/s10815-019-01620-9
3.Dahlhoff M, Camera E, Schäfer M, Emrich D, Riethmacher D, Foster A, Paus R, Schneider MR. Sebaceous lipids are essential for water repulsion, protection against UVB-induced apoptosis and ocular integrity in mice. Development. 2016 May 15;143(10):1823-31. doi: 10.1242/dev.132753
4.Kolbe T, Sheety S, Walter I, Palme R, Rülicke T. Impact of superovulation and mating on the wellbeing of juvenile and adult C57BL/6N mice. Reprod Fertil Dev. 2016 Jun;28(7):969-973. doi: 10.1071/RD14372
5.Dahlhoff M, Frances D, Kloepper JE, Paus R, Schäfer M, Niemann C, Schneider MR. Overexpression of Epigen during Embryonic Development Induces Reversible, Epidermal Growth Factor Receptor-Dependent Sebaceous Gland Hyperplasia. Mol Cell Biol. 2014 Aug 15;34(16):3086-95.
6.Kolbe T, Palme R, Touma C, Rülicke T. Repeated use of surrogate mothers for embryo transfer in the mouse. Biol Reprod. 2012 Jan 30;86(1):1-6. doi: 10.1095/biolreprod.111.092445
7.Dahlhoff M, Müller AK, Wolf E, Werner S, Schneider MR. Epigen Transgenic Mice Develop Enlarged Sebaceous Glands. J Invest Dermatol. 2010 Feb;130(2):623-6.
